Page 255 - Science Course 1 (Book 1)
P. 255
Mo6-L7c: What are Mixtures?
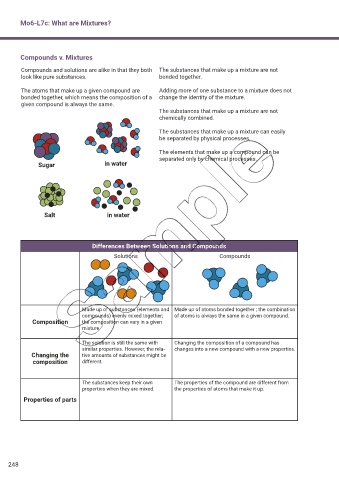
Compounds v. Mixtures
Compounds and solutions are alike in that they both The substances that make up a mixture are not
look like pure substances. bonded together.
The atoms that make up a given compound are Adding more of one substance to a mixture does not
bonded together, which means the composition of a change the identity of the mixture.
given compound is always the same.
The substances that make up a mixture are not
chemically combined.
The substances that make up a mixture can easily
be separated by physical processes.
The elements that make up a compound can be
separated only by chemical processes.
Sugar in water
Salt in water
Differences Between Solutions and Compounds
Solutions Compounds
Made up of substances (elements and Made up of atoms bonded together ; the combination
compounds) evenly mixed together; of atoms is always the same in a given compound.
Composition the composition can vary in a given
mixture.
The solution is still the same with Changing the composition of a compound has
similar properties. However, the rela- changes into a new compound with a new properties.
Changing the tive amounts of substances might be
composition different.
The substances keep their own The properties of the compound are different from
properties when they are mixed. the properties of atoms that make it up.
Properties of parts
248